Implant treatment has a well-established body of evidence supporting its long-term success and efficacy. Following in the shadow of this success however we now have the problem of peri-implantitis, characterised by inflammation and degeneration of the hard and soft tissues surrounding the implant and eventually leading to its loss from the jaw bone. Various techniques have been advocated to treat this infection taking its origins from periodontal treatment such as non-surgical, surgical and regenerative procedures.
The purpose this study was to systematically review the literature on the surgical regenerative treatment of the peri-implantitis and to determine an effective therapeutic predictable option for its clinical management.
Methods
The review followed the PRISMA statement (Moher et al. 2009) the protocol was also registered on the International Prospective Register of Systematic Reviews (PROSPERO) database. Searches were carried by two independent researchers using Ovid MEDLINE, PubMed, Embase, and Dentistry and Oral Sciences Source. Databases were searched from January 2006 to March 2016 and restricted to English, manual searches were also carried out in the relevant major journals. Inclusion criteria were: Human prospective and retrospective observational studies involving at least one surgical regenerative treatment method for peri-implantitis. Minimum sample size was 10 implants with no less than 12 months follow-up. Excluded studies included animal and in vitro studies, patients with uncontrolled systemic disease that put the implant at risk and ceramic or coated implants. Quality appraisal was carried out by two independent reviewers using the Cochrane Collaboration tool for assessing risk of bias in randomised trials (Higgins JPT et al. 2011)
Results
- From 883 records only 18 fulfilled the inclusion criteria. This included 8 prospective clinical studies, seven case series and three randomised clinical trials (RCT’s). A total of 528 patients with 713 implants were treated.
- 2 studies were at low risk of bias, 1 moderate, and 3 high. The remainder were classified as unclear.
| Total mean radiological bone level change | +2.97 mm | (95% CI 1.58 to 2.35) |
| · Mean radiological bone level + membrane | +1.86 mm | (95% CI 1.36 to 2.36) |
| · Mean radiological bone level – membrane | +2.12 mm | (95% CI 1.46 to 2.78) |
| · Mean radiological bone level submerged | +2.17 mm | (95% CI 1.87 to 2.47) |
| · Mean radiological bone level non-submerged | +1.91 mm | (95% CI 1.44 to 2.39) |
| Total mean probing depth change | -2.78 mm | (95% CI 2.31 to 3.25) |
| · Mean probing depth change + membrane | -2.88 mm | (95% CI 2.31 to 3.45) |
| · Mean probing depth change – membrane | -2.60 mm | (95% CI 1.90 to 3.30) |
| · Mean probing depth change submerged | -2.68 mm | (95% CI 1.71 to 3.64) |
| · Mean probing depth change non-submerged | -2.77 mm | (95% CI 2.23 to 3.30) |
| Total mean bleeding on probing change | -55% | (95% CI 45.2 to 64.4) |
Conclusions
The authors concluded: –
Within the limits of this systematic review, surgical regenerative treatment is a predictable option in managing peri-implantitis and improving clinical parameters of peri-implant tissues. There is no fundamental advantage of membrane use for bone graft coverage or submergence of the healing site on the final outcome of peri-implant defect regeneration. Due to the limited number of randomised clinical trials, at the time there is a lack of scientific evidence in the literature regarding the superiority of the regenerative versus non-regenerative surgical treatment
Comments
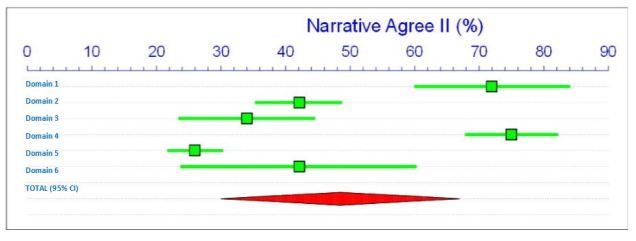
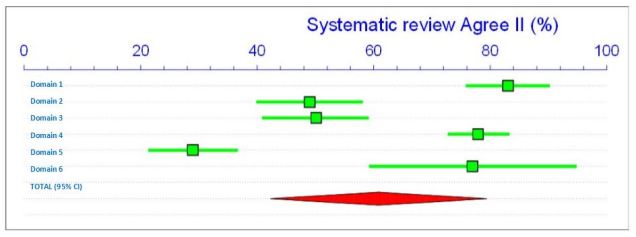
There are a few points to mention in relation to this well conducted review. Firstly, there is a lack of high quality studies with only 2 out of the 18 fulfilling the criteria and how this might influence the overall meta-analysis (this is commonly missed out in dental related systematic reviews, even though it is one of the PRISMA criteria the authors mention they adhere to). PRISMA has since been updated (Moher et al. 2015). Secondly, the risk of bias tool is designed for RCT’s (3/18) and there was no mention of using one of the tools specifically designed for non-randomised observational studies (Sterne et al. 2016; Wells 2013).
Finally, the author concludes that surgical regenerative treatment is a predictable option for the treatment of peri-implantitis but fails to mention how well or poorly this performs against standard non-regenerative debridement of the lesion. The reason I mention this is that a second paper was published by members of the same team, in the same institution, in the same month that could have shed some light on clinical effectiveness (Ramanauskaite et al. 2016).
First Posted on the National Elf Service
Links
Primary paper
Daugela P, Cicciù M, Saulacic N. Surgical Regenerative Treatments for Peri-Implantitis: Meta-analysis of Recent Findings in a Systematic Literature Review. J Oral Maxillofac Res. 2016 Sep 9;7(3):e15. eCollection 2016 Jul-Sep. Review. PubMed PMID: 27833740; PubMed Central PMCID: PMC5100640.
Other references
Original review protocol on PROSPERO
Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA; Cochrane Bias Methods Group; Cochrane Statistical Methods Group. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011 Oct 18;343:d5928.
Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016 Oct 12;355:i4919. doi: 10.1136/bmj.i4919. PubMed PMID: 27733354; PubMed Central PMCID: PMC5062054.
Wells, G.A., 2013. Newcastle Ottawa scale Coding Manual for Case-Control Studies. The Ottawa Hospital Research Institute.
Ramanauskaite A, Daugela P, Faria de Almeida R, Saulacic N. Surgical Non-Regenerative Treatments for Peri-Implantitis: a Systematic Review. J Oral Maxillofac Res. 2016 Sep 9;7(3):e14. eCollection 2016 Jul-Sep. Review. PubMed PMID: 27833739; PubMed Central PMCID: PMC5100639.
 With the advances in preventative dentistry one consequence has been patients becoming edentulous later in life. Accepting and adapting to conventional complete denture can be extremely challenging both physically and psychologically. There is now good evidence to support implant supported mandibular overdentures (Feine et al., 2002) using standard implants.
With the advances in preventative dentistry one consequence has been patients becoming edentulous later in life. Accepting and adapting to conventional complete denture can be extremely challenging both physically and psychologically. There is now good evidence to support implant supported mandibular overdentures (Feine et al., 2002) using standard implants.